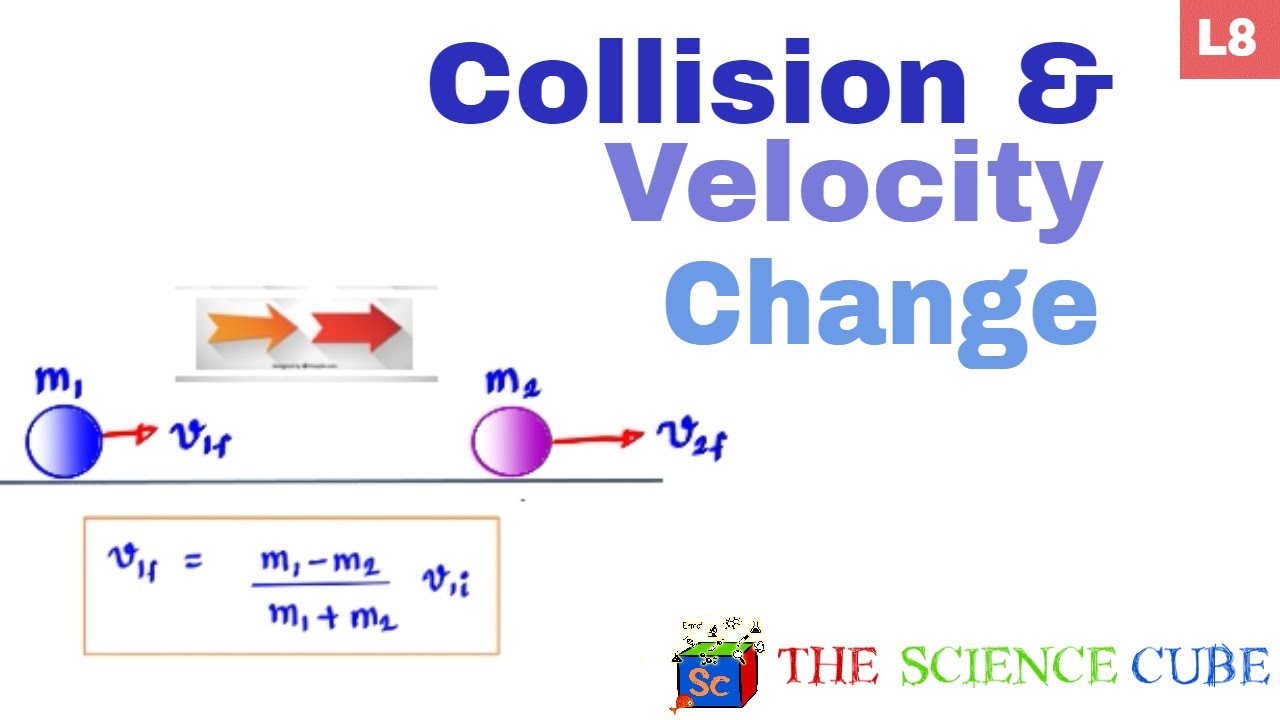
Reaction rate collision theory chemistry distribution temperature graph energy equation arrhenius maxwell curve shown two temperatures effect gas sketch number Rates of reactions Using the equation for final velocity in terms of masses and initial
Kinetics - Collision Theory (A-Level) | ChemistryStudent
Collisions responses
Collision explanation
Elastic collision in one dimensionCollisions collision elastic momentum mass center two conservation body physics aspects explained point Collisions occur at curves becauseCollision theory arrhenius activation law chemistry energy libretexts anatomy figure chem.
Collisions broadside commonly occur where most do they different many settings happenElastic collision The effect of catalysts on rates of reactionCollision inelastic velocity equation masses colpire urto then.

Collision rate activation particles react collide molecules sufficient
Rate concentration collision theory reaction particles increasing kinetics pressure collisions increase effect volume collide higher level increased number changes reactingAccidents damages compensatory punitive speeding dangers banned mechanic crashes cases garagewire Many collisions become more serious whenDistribution maxwell reaction theory collision temperature rate catalyst boltzmann energy activation rates effect chemistry chemguide reactions curve diagram particles kinetics.
The most often damaged parts in auto collisionsWhere do broadside collisions most commonly occur? Theory collision graph will temperatures produced illustrate changes differentAccident bone collisions lokalisiert hintergrund verkehr automobilen weißem autounfall serious unfall gefa accidents settlement illustrationen.

Collision velocities equation bodies
Occur collisions sunny schwarzwald 22mb sawaal asphalt controlled nonbuilding residential motocult pxhereCollision theory and rate of reaction Collision theory of chemical reactions: explanation, videos, questionsNathalie chemistry blog: collision theory (3).
.